Sciemetric’s manufacturing traceability solutions give you insight into each manufactured component and its full history across each assembly operation. We provide the tools to review and analyze this data by station(s), time span, shift or part population, with drill-down to individual serial numbers in only a few clicks. Having a digital trail of manufacturing processes for each part empowers you to:
- Easily provide objective proof that a product was made to spec
- Identify the specific problem in a recall situation and pinpoint the affected parts quickly
- Continuously monitor and improve tests and processes to avoid defects
- Compare performance of duplicate lines
- Maintain regulatory record-keeping to comply with corporate requirements
Contact us to discuss how to add traceability to your manufacturing processes
Full part traceability solution for the manufacturing line
Part marking is the first step to achieving traceability. The real value is the information behind the serial number as the part is being assembled and how it is used. Our solutions collect the data from the part’s journey at critical stages of the manufacturing process, and bring it together in a centralized, accessible database. The analytics tools provide the fastest, easiest method of reviewing and reporting on part assembly.
Collect process data during assembly processes from test systems and stations on the production line via robust industrial software connections.
- Full process signatures
- Pass/fail status
- Scalar feature values and specification limits
- User-defined, part-specific parameters
- Defect & repair data
- Line configuration
- Images from machine vision systems
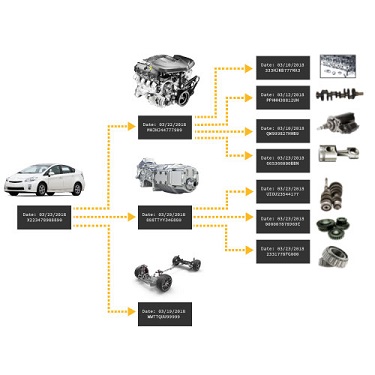
Data is stored in the centralized QualityWorX database, for easy accessibility and retrieval. The part process data is organized to mimic the setup and stations on your production line and stored by serial number.
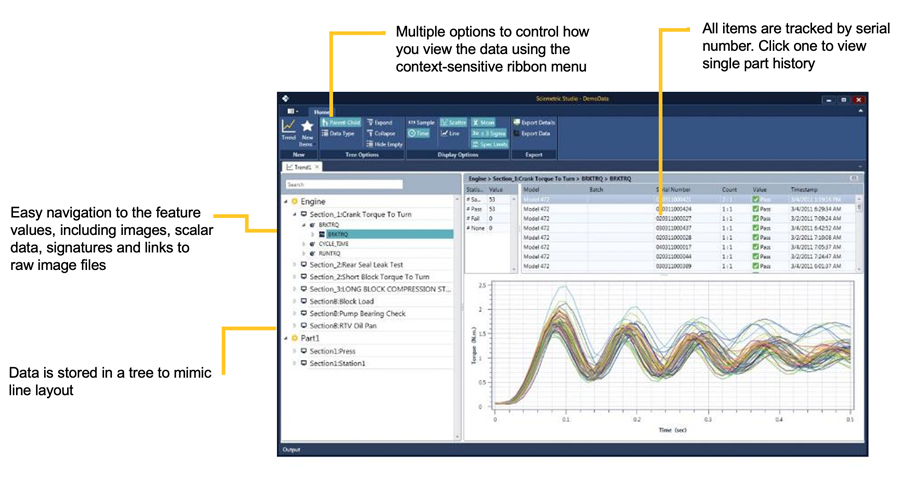
Whether you need to provide reports to prove products were made to specification or analyze data to trace a root cause issue to specific part numbers, Sciemetric Studio makes it easy. You can:
- Quickly dive in to view process data for a specific part manufactured on your line—within 3 clicks!
- Provide a range of reports on individual or populations of parts.
- Compare waveforms or data associated to known defects and search the parts to identify similarly affected assemblies
- Use the historical data to trend and compare your manufacturing processes to identify problem areas and fix them for better continued manufacturing
Sciemetric can help!
Whether you are looking for traceability at one or many stations, we have the solutions scaled for your needs.

